Accepted: 11 August 2024
Published: 24 August 2024
Babar Ijaz
Institute of Food Crops, Yunnan Academy of Agricultural Sciences, Kunming
650205, China
Correspondence: babarijazpbg@gmail.com
Plant diseases cause crop yield losses and compromise global food security, which makes the selection of disease-resistant cultivars a major goal of crop breeding. Quantitative disease resistance (QDR), regulated
by quantitative trait loci (QTLs) and considered to be durable and broad-spectrum resistance (BSR), has been a major breeding objective against various pathogen races. Immunity to plant diseases is mainly regulated
by pattern-triggered immunity (PTI), which is activated by pattern recognition receptors (PRRs) that detect pathogen-associated molecular patterns (PAMPs) or damage-associated molecular patterns (DAMPs) in
plants, respectively. In contrast effector-triggered immunity (ETI) is activated by nucleotide-binding leucine-rice proteins (NLRs) following a hypersensitive immune response.
Keywords: Wall associated kinases, disease resistance, Zea mays,
monocots.
Wall-associated receptor like kinases (WAKs)
Wall-associated receptor-like kinases or WAKs are a subgroup within the receptor like kinase (RLK) superfamily, extensively involved in cell wall elongation or tolerance to biotic and abiotic stresses; they are particularly renowned for their role in disease resistance. A recent study reported that 220,038 RLKs, dispersed across 300 plant genomes, illustrate the diversity of this gene class. WAKs constitute 11.1% of all
RLKs, making them one of the largest RLK families after LRR (44.0%) and G-LecRLK (13.9%) (Yin et al., 2024). WAKs are composed of an extracellular ligand-binding domain that interacts with pectin as cell surface receptors
activated by disease occurrence. They exhibit a transmembrane protein structure with domains that interact with extracellular effectors through a more conserved intracellular kinase domain. WAKs are also classified as arginine aspartate (RD-WAK) and non-RD-WAK based on the presence or absence of a conserved arginine and an adjacent catalytic aspartate residue in the kinase domain. Although earlier studies confirm that RD kinases play an efficient role
in plant growth and development, non-RD kinases are important for disease resistance (Stephens et al., 2022).
However, there are some recent reports that RD-WAKs also play critical roles in disease resistance.
WAKs: Role in Plant Defense against diseases.
WAKs control a variety of pathogen hosts, including race-specific, extracellular and intracellular and pathogen attack on single and multiple plant species (Stephen et al., 2022). In Arabidopsis, the AtWAK1 gene enhanced resistance to Botrytis cinerea, confirming their role in plant immunity. AtWAKL22-RFO1 is required for resistance against Fusarium oxysporum 1 (RFO1). WAKL22-ROF1 activates the Mitogen-Activated Protein Kinase (MAPK) signaling cascade and senses pectin methylation, providing early defense against the pathogen (Huerta et al., 2023).
Race-specific resistance to Leptosphaeria maculans and Zymoseptoria tritici is conferred by WAKL genes Rlm9 and Stb6 in Brassica and wheat, respectively (Larkan et al., 2020;
Saintenac et al., 2018). Xanthomonas oryzae (Xoo) is managed by a race-specific, durable resistance WAK gene, Xa4, which modulates the CesA expression to facilitate cellulose biosynthesis and provide resistance against Xoo, and is also involved in cell wall enforcement (Hu et al., 2017). For chitin-induced responses, GhWAK7A interacts with chitin receptors and activates a signaling system against Verticillium dahliae (Vd) and Fusarium oxysporum in cotton (Wang et al., 2020). Additionally, GhWAK7 is not involved in oligogalacturonides (OG) responses, as it does not affect MAPK activation or ROS production.
In maize (Zea mays), early studies identified different Ht loci (Ht1, Ht2 and Ht3), with the Htn1 gene providing resistance against northern corn leaf blight (NCLB). It was later found to encode a WAK-associated protein (ZmWAK-RLK1), which is involved in delayed lesion formation of NCLB (Yang et al., 2021). Another gene ZmWAK/qHSR1, highly expressed in the maize mesocotyl, confers quantitative resistance against head smut by promoting the salicylic acid pathway (Zuo et al., 2015). WAKs that confer disease resistance through cell wall modification also modifies composition of the cell wall as an additional role, with an increase in strength of cell wall to prevent pathogen penetration during disease occurrence. ZmWAK/qHSR1 is involved in cell turgor regulation and osmotic stress tolerance, which promotes cell growth, whereas, in rice, Xa-mediated resistance to Xoo increases the expression of CesA genes, leading to increased mechanical strength.
WAK-mediated GLS disease resistance in maize
Gray leaf spot (GLS) is one of the most important foliar diseases caused by the fungal pathogens Cercospora zeae-maydis and Cercospora zeina. Commonly occurring on mature leaves, it can cause 70-100% of yield losses. Previous investigations concluded that GLS as a quantitative trait, and although numerous QTL studies have been reported, only a few genes have been cloned for GLS resistance. Recently, two consecutive studies highlighted the role of WAK-mediated quantitative resistance to GLS in maize.
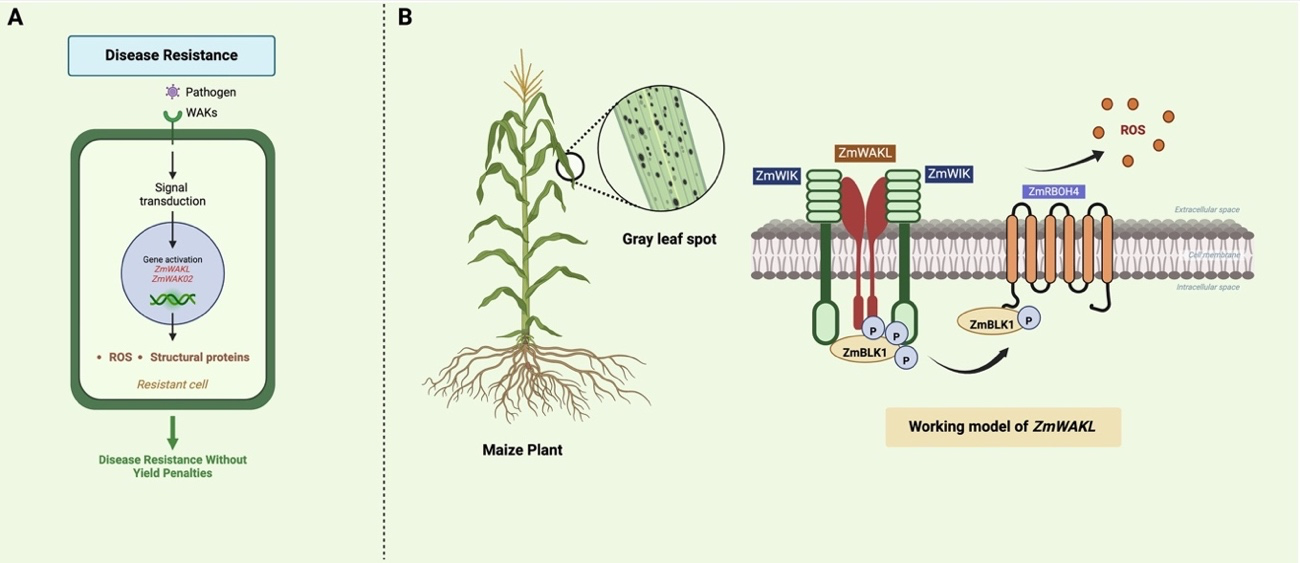
Zhong et al. (2024) described ZmWAKL, a gene encoding a cell wall-associated kinase-like protein, which works as a pattern recognition receptor. It was identified through sequential genetic mapping from the populations derived from Y32 (a GLS-resistant line) and Q11 (a GLS-susceptible line). They fine-mapped a 60-kb QTL region in Y32 with three genes, including ZmWAKLY, which encodes protein to exhibit self-association and kinase activity and is localized to the plasma membrane. Over-expression of ZmWAKLY increased GLS resistance induced by C. zeina infection (Zhong et al., 2024). Further investigation of ZmWAKL revealed its coreceptor, ZmWIK, which is also localized to the plasma membrane and exhibits kinase activity (Figure 1B). The ZmWAKLY/ZmWIK complex interacts with ZmBLK1 to initiate immune signaling upon pathogen attack, which later interacts with ZmRBOH4 to phosphorylate and produce extracellular reactive oxygen species (ROS), causing quantitative resistance to the disease.
Dai et al. (2024) identified a major QTL (qRglsSB) that explains 58.42% of the phenotypic variance in the BC1F1 population, with three significant candidate genes (ZmWAK02, ZmWAK03, ZmWAK04) encoding RD-WAK domains chosen from twelve genes identified within an 817-kb QTL region (Dai et al., 2024). Transgenic lines of these three WAK genes confirmed that only ZmWAK02 is responsible for strong GLS resistance in maize, compared with ZmWAK03 and ZmWAK04. ZmWAK02 is considered to be of great agronomic importance in maize molecular breeding, showing a large effect and strong resistance to GLS for both pathogenic agents. The introgression of ZmWAK02 didn’t compromise maize yield under GLS pressure and caused no change in yield, plant height, days to tassel, days to silking, days to anthesis, and ear height of the crop.
Concluding remarks
The expansion of WAKs across a wide range of plant species has opened new directions to develop WAK-mediated pathogen-resistant crops (Figure 1A). Quantitative disease resistance (QDR) in pathogen immunity represents a multifaceted aspect of plant defense mechanisms, influenced by the cumulative effects of multiple genetic factors and environmental conditions. The immune module of WAKL–WIK–BLK1–RBOH4 exhibits the regulation of QDR genes like ZmWAKL, which initiate defense responses against GLS in maize (Zhong et al., 2024). Apart from non-RD-WAKs, the RD-WAKs (ZmWAK02) also provide additional insights into disease resistance without yield penalties, although the further mechanisms is unknown. Furthermore, the development of the RLKdb database (https://biotec.njau.edu.cn/rlkdb/) provides annotation information for more than 300 plant genomes, which will be beneficial for genome-wide and transcriptome-wise classification of WAKs as well as for evolutionary and functional studies (Yin et al., 2024).
Previously, most work on disease resistance focused solely on the molecular mechanisms, lacking their use in plant resistance breeding. The discovery of QDR genes offers a huge opportunity to introgress these genes into parental lines of hybrids and develop resistant varieties via marker-assisted selection (MAS) approach. WAKs genes showed substantial improvements in resistance to head smut and gray leaf spot in maize with the introgression of ZmWAK/qHSR1 and ZmWAK02, respectively (Zuo et al., 2015; Dai et al., 2024). Therefore, the characterization of WAKs will serve as a model for the cloning of disease resistance QTLs, followed by gene function studies of large-effect loci in gene introgression, thereby creating disease resistance in crops. QDR acquired through WAKs is a valuable resource for breeding disease-resistant crop varieties without yield penalties.
Further research should explore additional components of the plant immune system, including the cross-talk and interaction between WAKs and the mechanism by which WAKs discern and transduce specific pathogenic signals. The functions of RD-WAKs and the difference between the molecular mechanisms of RD-WAKs and non-RD WAKs should also be explored. Additionally, exploring the genetic and epigenetic regulation of WAKs could unveil new strategies for improving crop resilience against pathogens through biotechnological and breeding approaches.
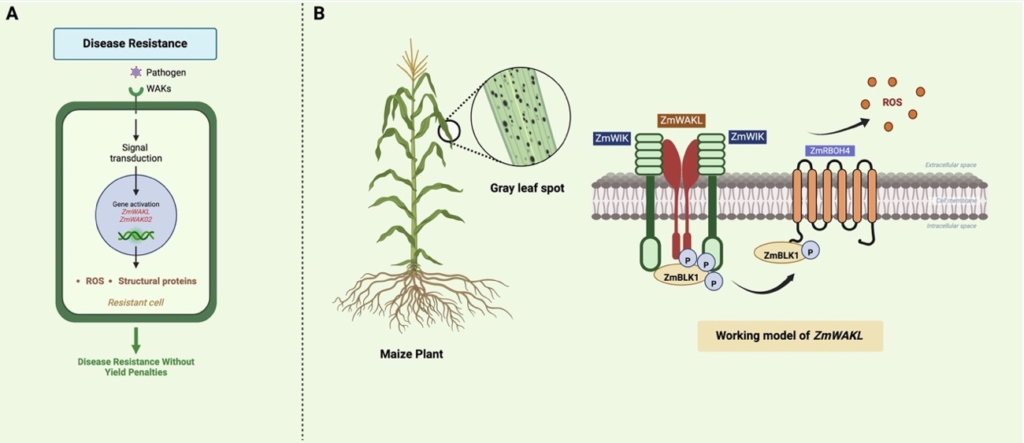
Figure. 1 | (A) Schematic diagram illustrating WAK-mediated disease resistance without yield penalties. (B) Working model of ZmWAKLY for resistance against gray leaf spot (GLS). The pathogen attack increases the levels of phosphorylation of ZmWAKLY, ZmBLK1 and ZmRBOH4, and triggers ROS burst to cause GLS resistance in Maize.
Acknowledgments: We apologize to the relevant works not cited due to space limitations. No conflict of interest is declared.
Conflict of Interest: None.
References
- Dai, Z., Pi, Q., Liu, Y., Hu, L., Li, B., et al. (2024). ZmWAK02 encoding an RD-WAK protein confers maize resistance against gray leaf spot. New Phytol. 241:1780-1793.
- Hu, K., Cao, J., Zhang, J., Xia, F., Ke, Y., Zhang, H., Xie, W., et al. (2017). Improvement of multiple agronomic traits by a disease resistance gene via cell wall reinforcement. Nature Plants 3:17009.
- Huerta, AI., Sancho-Andrés, G., Montesinos, J.C., Silva-Navas, J., Bassard, S., Pau-Roblot, C., et al. (2023). The WAK-like protein RFO1 acts as a sensor of the pectin methylation status in Arabidopsis cell walls to modulate root growth and defense. Mol. Plant. 16:865-881.
- Larkan, N.J., Ma, L., Haddadi, P., Buchwaldt, M., Parkin, I.A.P., Djavaheri, M., and Borhan, M.H., (2020) The Brassica napus wall-associated kinase-like (WAKL) gene Rlm9 provides race-specific blackleg resistance. Plant J. 104:892-900.
- Saintenac, C., Lee, W.S., Cambon, F., Rudd, J.J., King, R.C., Marande, W., Powers, S.J., et al. (2018). Wheat receptor-kinase-like protein Stb6 controls gene-for-gene resistance to fungal pathogen Zymoseptoria tritici. Nat. Genet. 50:368-374.
- Stephens, C., Hammond-Kosack, K.E., and Kanyuka K. (2022). WAKsing plant immunity, waning diseases. J. Exp. Bot. 73:22-37.
- Wang, P., Zhou, L., Jamieson, P., Zhang, L., Zhao, Z., Babilonia, K., Shao, W., et al. (2020). The Cotton Wall-Associated Kinase GhWAK7A Mediates Responses to Fungal Wilt Pathogens by Complexing with the Chitin Sensory Receptors. Plant Cell 32:3978-4001.
- Yang, P., Scheuermann, D., Kessel, B., Koller, T., Greenwood, J.R., Hurni, S., et al. (2021) Alleles of a wall-associated kinase gene account for three of the major northern corn leaf blight resistance loci in maize. Plant J. 106:526-535.
- Yin, Z., Liu, J., and Dou, D., (2024). RLKdb: A comprehensively curated database of plant receptor-like kinase families. Mol. Plant. doi: 10.1016/j.molp.2024.02.014.
- Zhong, T., Zhu, M., Zhang, Q., Zhang, Y., Deng, S., Guo, C., et al. (2024). The ZmWAKL-ZmWIK-ZmBLK1-ZmRBOH4 module provides quantitative resistance to gray leaf spot in maize. Nat. Genet. 56:315-326.
- Zuo, W., Chao, Q., Zhang, N., Ye, J., Tan, G., Li, B., Xing, Y., Zhang, B., et al. (2015). A maize wall-associated kinase confers quantitative resistance to head smut. Nat. Genet